Subclass Pterygota
 About the same time primitive insects began to diversify and exploit terrestrial habitats, other types of arthropods (especially spiders and scorpions) were also invading the land. Almost all of these arachnids were predatory, and it seems likely that insects were a major component of their diet. If this is true, then the selective pressure of predation may explain why most surviving members of the Apterygota are cryptozoic (adapted for life in concealed places — in leaf litter, under bark, etc.).
About the same time primitive insects began to diversify and exploit terrestrial habitats, other types of arthropods (especially spiders and scorpions) were also invading the land. Almost all of these arachnids were predatory, and it seems likely that insects were a major component of their diet. If this is true, then the selective pressure of predation may explain why most surviving members of the Apterygota are cryptozoic (adapted for life in concealed places — in leaf litter, under bark, etc.).
Predation also may have been the driving force behind an event that is probably the single most significant landmark in the evolutionary history of insects — development of wings. Flight was not only the ultimate escape from earth-bound predators, but it also proved to be a rapid form of transportation, an efficient mode of dispersal, and a convenient way to find a mate!! Insects were the first living organisms to master flight, and for more than 150 million years (until the first flying reptiles appeared in the Jurassic period) the insects were undisputed rulers of the air.
How, when, and where did wings evolve? These questions have puzzled entomologists since the days of Charles Darwin. The fossil record is not very informative because fully winged species appeared rather suddenly in the Carboniferous period (about 380 million years ago) with no clear evidence of ancestral precursors. Like all other adaptations, the development of functional wings must have been influenced by the process of natural selection acting over a span of many generations. But what adaptive value could there have been for protowings that were too small for sustained flight?
Some biologists have speculated that wings may have developed as modifications of tracheal gills in aquatic insects (the gill theory), from flaps of integument covering thoracic spiracles (the spiracular flap theory), from pre-existing appendages near the legs (the endite-exite theory), or as lateral extensions of dorsal plates on the thorax (the paranotal lobe theory). These structures were not necessarily related to flight — they may have played a role in sexual selection (helping to attract a mate), in thermal regulation (absorbing heat energy like a passive solar panel), or in aerodynamics (improving the stability of a jumping or gliding insect). Any of these theories could account for the development of large wing-like structures, but none have been widely embraced because they all fail to explain how or why such appendages acquired a hinge-joint mechanism, strong thoracic musculature, and the neural complexity necessary for sustained flight. The debate continues, but so far it has produce more heat than light.
 In 1994, James Marden and Melissa Kramer proposed an attractive new theory that may add fresh insight to the puzzle of wing evolution. They suggested flight may have originated among aquatic insects living on the water surface. Such insects could have used their gills or gill covers as oars to row across the water. Selective pressure for a more efficient rowing mechanism could lead to larger oars (protowings) and stronger muscles. High speed rowing movements might easily progress into skimming along the water surface, and eventually to self-powered flight. In fact, surface-skimming behavior has been observed in both mayflies (Ephemeroptera) and stoneflies (Plecoptera), and there is direct experimental evidence of a correlation between wing size (surface area) and skimming velocity in Taeniopteryx burksi, a small winter stonefly with a feeble mastery of flight. Marden and Kramer’s theory is noteworthy because it accounts for the progressive development of wings as well as the muscles needed to power them.
In 1994, James Marden and Melissa Kramer proposed an attractive new theory that may add fresh insight to the puzzle of wing evolution. They suggested flight may have originated among aquatic insects living on the water surface. Such insects could have used their gills or gill covers as oars to row across the water. Selective pressure for a more efficient rowing mechanism could lead to larger oars (protowings) and stronger muscles. High speed rowing movements might easily progress into skimming along the water surface, and eventually to self-powered flight. In fact, surface-skimming behavior has been observed in both mayflies (Ephemeroptera) and stoneflies (Plecoptera), and there is direct experimental evidence of a correlation between wing size (surface area) and skimming velocity in Taeniopteryx burksi, a small winter stonefly with a feeble mastery of flight. Marden and Kramer’s theory is noteworthy because it accounts for the progressive development of wings as well as the muscles needed to power them.
Based on the comparative morphology of wing structure, venation, and flight mechanics, it appears that the ability to fly evolved only once in the class Insecta. All remaining orders are grouped into a single subclass, the Pterygota (from the Greek word “pterygo” meaning a wing) because they are believed to have all descended from winged ancestors. Some pterygote insects (lice and fleas, for example) have acquired a specialized lifestyle where wings have been lost because they provided no selective advantage. These insects are said to be “secondarily wingless” — their winglessness is a derived adaptation (apomorphic condition) not to be confused with the primitively wingless state (pleisiomorphic condition) of apterygote insects.
 Concurrent with the development of wings, the first pterygote insects also show gradual developmental changes in body form as they mature. Wings and external genitalia grow and develop during the immature stages, becoming complete and functional only after the final molt into adulthood. This gradual change in body form is called hemimetabolous development (incomplete metamorphosis). Immature stages of hemimetabolous insects are known as nymphs (or naiads if they are aquatic) rather than young (as in ametabolous insects).
Concurrent with the development of wings, the first pterygote insects also show gradual developmental changes in body form as they mature. Wings and external genitalia grow and develop during the immature stages, becoming complete and functional only after the final molt into adulthood. This gradual change in body form is called hemimetabolous development (incomplete metamorphosis). Immature stages of hemimetabolous insects are known as nymphs (or naiads if they are aquatic) rather than young (as in ametabolous insects).
For descriptive purposes, all insects with hemimetabolous development are grouped together as Exopterygota. This name (from the Greek “exo” meaning outer and “pterygo” meaning wing) reflects the fact that wings develop externally as outgrowths of the nymph’s body wall. They grow larger with each molt and usually become visible as stationary “wing pads” on the top or sides of the thorax. Functional wings occur only in adult insects.
The Paleopterous Orders
 The most primitive winged, hemimetabolous insects have large, membranous wings that are held continuously out to the side of the body or over the back of the thorax. These insects do not have the ability to fold their wings down flat over the abdomen. All orders that share this “primitive” condition are grouped as Paleoptera, a subdivision (infraclass) within the subclass Pterygota. The word paleoptera is derived from the Greek words “paleo-” meaning primitive, and “ptera” meaning wings.
The most primitive winged, hemimetabolous insects have large, membranous wings that are held continuously out to the side of the body or over the back of the thorax. These insects do not have the ability to fold their wings down flat over the abdomen. All orders that share this “primitive” condition are grouped as Paleoptera, a subdivision (infraclass) within the subclass Pterygota. The word paleoptera is derived from the Greek words “paleo-” meaning primitive, and “ptera” meaning wings.
Based largely on the fossil record, entomologists now recognize up to seven orders of paleopterous insects. Most of these orders were relatively abundant during the Carboniferous period, but they disappeared suddenly in the Permian (about 250 million years ago). The largest insect that ever lived, Meganeura monyi Brongniart, was a member of one of these extinct orders, the Protodonata. It was a damselfly-like insect with a body length of 30 cm (1 foot) and a wingspan of nearly 75 cm (2.5 feet).
Of all the paleopterous orders, only two (Ephemeroptera and Odonata) have survived to the present time. Immatures of both orders are exclusively aquatic (naiads = water nymphs). Adults are predominantly aerial: their legs are often reduced or adapted for clinging or grasping, never for walking or running. Adaptations for internal fertilization (penis-like structures) first appeared in adults of the Paleoptera.
Ephemeroptera
 The Ephemeroptera (or Ephemerida) are called mayflies, but may also be known locally as shadflies, lakeflies, or spinners. Immatures are aquatic; they generally live in unpolluted habitats with fresh, flowing water. Some species are active swimmers, others are flattened and cling to the underside of stones, a few are burrowers who dig U-shaped tunnels in the sand or mud. Most species are herbivorous. Their diet consists primarily of algae and other aquatic plant life scavenged from surrounding habitat. Some species mature quickly, in as little as four weeks, while others develop more slowly (one to four years per generation).
The Ephemeroptera (or Ephemerida) are called mayflies, but may also be known locally as shadflies, lakeflies, or spinners. Immatures are aquatic; they generally live in unpolluted habitats with fresh, flowing water. Some species are active swimmers, others are flattened and cling to the underside of stones, a few are burrowers who dig U-shaped tunnels in the sand or mud. Most species are herbivorous. Their diet consists primarily of algae and other aquatic plant life scavenged from surrounding habitat. Some species mature quickly, in as little as four weeks, while others develop more slowly (one to four years per generation).
 Once a mayfly completes development as a naiad, it leaves the aquatic environment, often rising to the water surface in a bubble of air. It quickly molts to a winged form (the subimago) and flies to a nearby leaf or stem. The subimago is a transitional stage. Within a few hours, it molts again into an imago, a sexually mature adult. The imago usually has transparent wings and a smooth, shiny exoskeleton in contrast to the cloudy wings and dull, pubescent body of the subimago. Mayflies are the only insects that molt again after they have wings.
Once a mayfly completes development as a naiad, it leaves the aquatic environment, often rising to the water surface in a bubble of air. It quickly molts to a winged form (the subimago) and flies to a nearby leaf or stem. The subimago is a transitional stage. Within a few hours, it molts again into an imago, a sexually mature adult. The imago usually has transparent wings and a smooth, shiny exoskeleton in contrast to the cloudy wings and dull, pubescent body of the subimago. Mayflies are the only insects that molt again after they have wings.
Most adults are delicate insects with a very short lifespan. More about
Ephemeroptera They do not feed (mouthparts are vestigal), and some species emerge, reproduce, and die in a single day. Males generally fly in swarms that undulate in the air 5-15 meters above the ground. Females fly into the swarm and are quickly grabbed by a male. Copulation takes place in flight, and the female usually lays her clutch of eggs within minutes or hours. Males die shortly after mating, females usually die soon after oviposition.
Odonata
 Odonata, dragonflies and damselflies, are predaceous both as immatures and adults. The adults are quick, agile fliers that are generally considered beneficial because they feed on large numbers of small, flying insects like gnats and mosquitoes. Legs are used either as a basket for catching prey or as grapples for clinging to emergent vegetation. Eggs are laid singly in fresh water; females often hover over open water and dip their abdomen as they oviposit.
Odonata, dragonflies and damselflies, are predaceous both as immatures and adults. The adults are quick, agile fliers that are generally considered beneficial because they feed on large numbers of small, flying insects like gnats and mosquitoes. Legs are used either as a basket for catching prey or as grapples for clinging to emergent vegetation. Eggs are laid singly in fresh water; females often hover over open water and dip their abdomen as they oviposit.
 Eggs hatch into aquatic immatures (naiads) that feed opportunistically on other forms of aquatic life including mayfly naiads, small crustaceans, annelids, and molluscs. Some of the larger dragonfly naiads will even attack small fish and tadpoles. All immature odonata have a specialized labium for catching prey. Folded under the head and thorax when not in use, the labium can be extended rapidly toward potential prey. Hooked lobes at the tip of the labium grasp or impale the prey and draw it back to the mouth as the labium retracts.
Eggs hatch into aquatic immatures (naiads) that feed opportunistically on other forms of aquatic life including mayfly naiads, small crustaceans, annelids, and molluscs. Some of the larger dragonfly naiads will even attack small fish and tadpoles. All immature odonata have a specialized labium for catching prey. Folded under the head and thorax when not in use, the labium can be extended rapidly toward potential prey. Hooked lobes at the tip of the labium grasp or impale the prey and draw it back to the mouth as the labium retracts.
Damselfly naiads are usually more slender than dragonfly naiads, and have three leaf-like gills at the end of the abdomen. More about
Odonata Dragonfly gills are located internally, within the rectum, where bellows-like contractions of the rectal muscles cause oxygenated water to circulate in and out.
The Neopterous Orders
The large, membranous wings of the Paleoptera were not always an advantage. The insects had to land occasionally to rest, feed, or mate, and once on the ground, big bulky wings would only attract the attention of predators and hinder movement through dense foliage. If the wings became broken or shredded by objects in the environment, they would be a major liability. It is probably worth noting that Ephemeroptera and Odonata, the only surviving members of the Paleoptera, do not live on the ground. Wings are their only means of locomotion; legs are used only for grasping or clinging. The adults of both orders have an aerial lifestyle that puts them beyond the reach of earth-bound predators.
It seems likely, therefore, that selective pressures on the first winged insects heavily favored the development of some mechanism for folding the wings against the body after landing, making them less conspicuous, less awkward, and less susceptible to breakage. Such an adaptation first appeared 350-400 million years ago (in the late Devonian or early Carboniferous period). The progeny of these wing-folders are placed in the infraclass Neoptera, meaning “new wing”. These insects represent a remarkably successful lineage; they became the ancestors of all “higher” orders of insects.
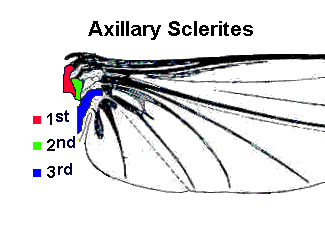 The “new wing” was articulated at its base so it could be folded flat over the insect’s abdomen when not in use. The folding mechanism is facilitated by an elastic hinge and a single flexor muscle attached to a small plate (third axillary sclerite) at the base of the wing. With this adaptation, winged insects could be as agile on the ground as they were in the air. Relative freedom from predation plus an abundant food supply (in the form of green plants) gave these winged insects a unique opportunity to exploit new ecological niches. Indeed, there was an unprecedented explosion of species during the upper Carboniferous period — at least eleven new insect orders appeared within a span of less than 30 million years.
The “new wing” was articulated at its base so it could be folded flat over the insect’s abdomen when not in use. The folding mechanism is facilitated by an elastic hinge and a single flexor muscle attached to a small plate (third axillary sclerite) at the base of the wing. With this adaptation, winged insects could be as agile on the ground as they were in the air. Relative freedom from predation plus an abundant food supply (in the form of green plants) gave these winged insects a unique opportunity to exploit new ecological niches. Indeed, there was an unprecedented explosion of species during the upper Carboniferous period — at least eleven new insect orders appeared within a span of less than 30 million years.
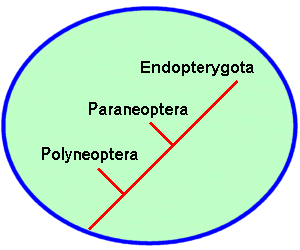 During this burst of adaptive radiation, the class Insecta split into at least three distinctive branches. Each branch represents a group of orders that share structural or developmental similarities, and presumably, arose from a common ancestor. Insects in the first branch, the Polyneoptera, are relatively unspecialized. They all have chewing mouthparts and incomplete (hemimetabolous) development. A second branch, the Paraneoptera, also includes hemimetabolous insects but their mouthparts show varying degrees of specialization for scraping, rasping, or piercing/sucking. The third branch encompasses all insects that undergo complete metamorphosis (holometabolous development). These insects have four stages in their life cycle: egg, larva, pupa, and adult. Systematists group all holometabolous insects as Endopterygota (from the Greek “endo” meaning inner and “pterygo” meaning wing). This name reflects the fact that wing buds appear only during the pupal stage, arising internally from embryonic tissues.
During this burst of adaptive radiation, the class Insecta split into at least three distinctive branches. Each branch represents a group of orders that share structural or developmental similarities, and presumably, arose from a common ancestor. Insects in the first branch, the Polyneoptera, are relatively unspecialized. They all have chewing mouthparts and incomplete (hemimetabolous) development. A second branch, the Paraneoptera, also includes hemimetabolous insects but their mouthparts show varying degrees of specialization for scraping, rasping, or piercing/sucking. The third branch encompasses all insects that undergo complete metamorphosis (holometabolous development). These insects have four stages in their life cycle: egg, larva, pupa, and adult. Systematists group all holometabolous insects as Endopterygota (from the Greek “endo” meaning inner and “pterygo” meaning wing). This name reflects the fact that wing buds appear only during the pupal stage, arising internally from embryonic tissues.

